- New
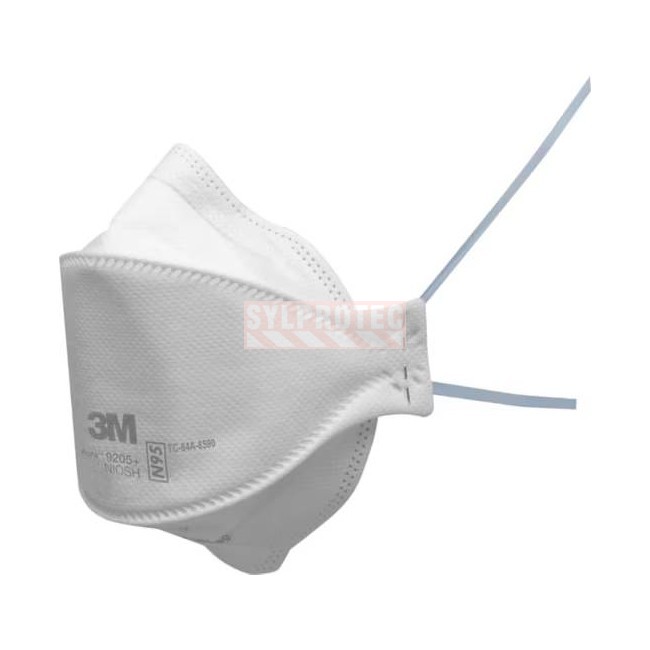



3M Aura 9205+ N95 respirator, NIOSH compliant, valveless, three-panel design with nose clip and nose foam. Ideal for healthcare, decontamination, and pandemic use. Case of 440 units.
No returnable if used. For more information about our return policy, please refer to the Orders and Returns.
The 3M Aura 9205+ particulate respirator is a NIOSH N95 compliant device designed to filter at least 95% of non-oil-based airborne particles. Its three-panel fold-flat design provides excellent face adaptability, increased breathing space, and superior comfort during extended wear.
3M Manufacturer Code: 7100232940, model: 9205+
The integrated M-noseclip and nose foam make it easy to seal around the nose and reduce air leakage on the sides. The advanced electrostatic filter media delivers high filtration efficiency while maintaining low breathing resistance, making it particularly suited for long-duration interventions.
Each respirator is individually packaged and sold in a case of 440 units, simplifying inventory management in hospital, industrial, or institutional settings. This case format is ideal for healthcare facilities, decontamination teams, emergency preparedness organizations, and high-consumption industrial operations.
The 9205+ Aura has no exhalation valve, making it an appropriate choice when environmental protection is required in addition to operator protection, such as in healthcare settings or pandemic response interventions.
Healthcare and hospital settings: the 9205+ meets N95 requirements recommended for protection against biological aerosols in clinical environments. It is used for patient care in respiratory illness contexts, aerosol-generating procedures, and respiratory isolation interventions.
Pandemic management and public health emergencies: its valveless design simultaneously protects the wearer and the surrounding environment. Emergency preparedness organizations include it in strategic stockpiles for rapid large-scale response.
Decontamination and site remediation: when working with biological agents, toxic dusts, or fine particles, the 9205+ provides the N95 filtration barrier required by decontamination protocols.
High-particle industries: construction, mining, grinding, sanding, mineral and wood processing: activities that generate non-oil-based particles for which N95 class protection is indicated.
Pharmaceutical manufacturing and food processing: the valveless two-way protection suits environments where cross-contamination must be prevented, such as cleanroom packaging and processing operations.
The 9205+ is compliant with NIOSH 42 CFR 84 for the N95 class. In Canada, its use fits within a respiratory protection program compliant with CSA Z94.4. In the United States, the program must comply with OSHA 1910.134, including fit testing, medical evaluation, and user training.
The three-panel three-dimensional shape creates space between the mask and the mouth, reducing heat and moisture build-up during extended wear. The braided polyisoprene straps provide consistent elasticity and do not irritate skin during repeated use.
The soft nose foam improves nasal comfort and contributes to better facial sealing, even across varying face shapes. These features make the 9205+ one of the most wearable N95 respirators available for long work shifts.
The case of 440 individually packaged units simplifies distribution, traceability, and inventory management. The 60-month useful life from the manufacturing date allows building a strategic stockpile without risk of rapid expiry.
The 9205+ and the 9210 are both 3M NIOSH N95 compliant respirators, but they differ in several ways. The 9205+ (model R9205) is sold in a case of 440 units, making it particularly suited for institutional orders and large-scale inventory management. The 9210 (model R9210) is sold individually and is better suited for occasional purchases or limited needs. In terms of protection, both models offer N95 filtration efficiency and have no exhalation valve.
The 9205+ is designed for limited use and must be removed and set aside if contamination, visible damage, deformation, increased breathing resistance, or seal failure is noted. In healthcare settings, institutional protocols and public health authority guidelines define the conditions for extended use or limited reuse. In industrial settings, the respirator is for single use per work conditions and employer policy.
Yes. The 9205+ is compliant with NIOSH N95, which is the recognized standard for respiratory protection against airborne biological agents. Its use in Canadian hospital settings must be part of a respiratory protection program compliant with CSA Z94.4, including fit testing, medical evaluation, and user training. Sylprotec can guide your facility toward the appropriate resources for program compliance.
For complete personal protection in hospital, decontamination, or industrial settings, recommended personal protective equipment includes latex 5 mil powder-free gloves (G555), Virtua Max 11872 clear lens safety eyewear with foam gasket (YA11872), EARSOFT 311-4106 detectable earplugs (OA4106), the flame-resistant SMS disposable coverall (V50) and the Tychem 2000 yellow coverall with hood (VQC127SYL). These products are available under the "Accessories" section at the bottom of the page.
Among comparable disposable respirators available at Sylprotec, the 3M 9210 respirator (R9210), the 9211 model with Cool Flow valve (R9211) and the 3M 8210 respirator (R8210) are complementary options depending on volume and usage context.
Data sheet
You might also like